Alternative Transportation Fuels
We don't have to compromise when using cleaner alternative fuels.

The 534 HP Aston Martin running on 60% bio-methanol, 40% gasoline at the British round of the Dutch Supercar
Challenge at Donington Park, UK, 7-8 May.
1. Introduction
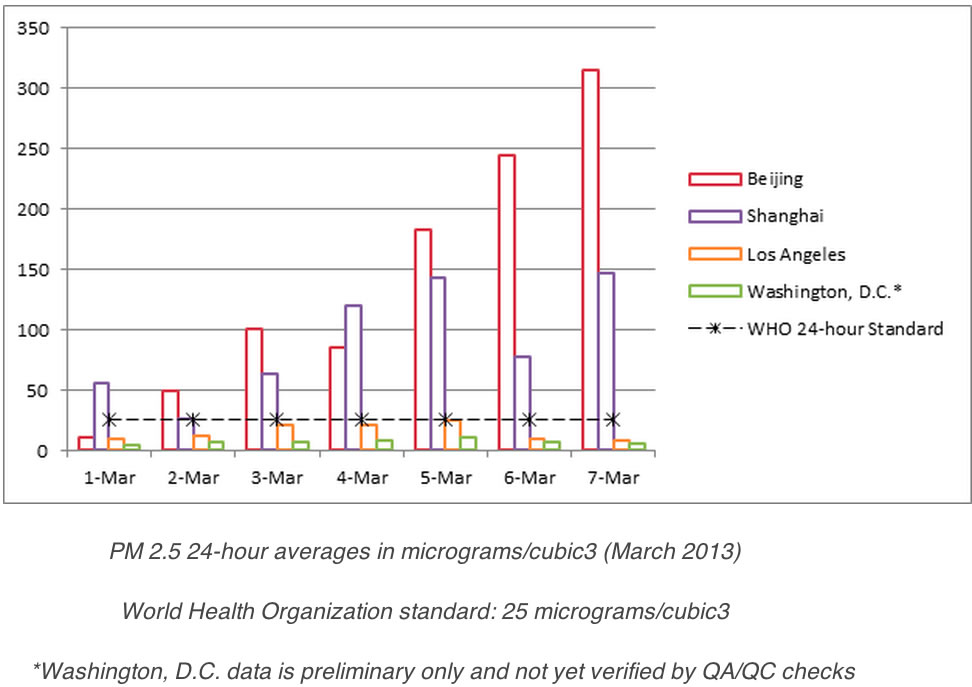
Air pollution is a major reason to move to alternative fuels. The chart below shows 24 hour averages of particulates in Beijing, Shanghai and Los Angeles. Although we know LA is bad, it is hard to imagine the quality of air in these Chinese cities.
Our need to reduce particulates (and other pollutants) will lead us to alternative fuels that simply burn cleaner. Three significantly different transportation solutions are likely to be long term alternatives. We will be using Internal Combustion Engines (ICE) for decades to come, primarily because we can envision thermodynamic efficiencies of the order of 50%. This is at the lower end of the range that fuel cells can achieve. Thus, ICE will hang in there for much longer than most people think. They will power the transportation solutions of plug-in hybrids, methanol/ethanol/gasoline mixtures, and natural gas vehicles. In time fuel cells will become reliable and cheap enough to replace them. When this happens we may achieve efficiencies of 75%.
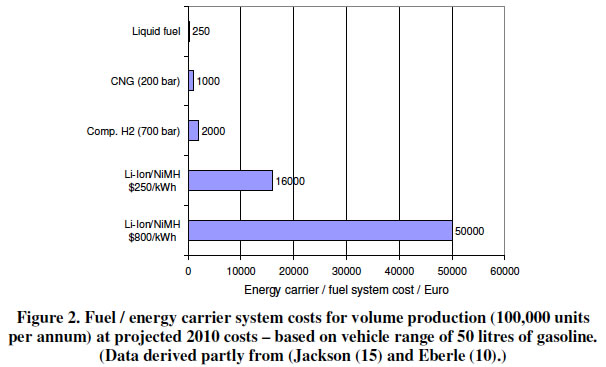
A comparison of the fuel/energy costs is shown below. It clearly shows that use of an alternative liquid fuel (top bar) is the most cost effective way of changing from oil.
2. The current Greenhouse Gas Situation
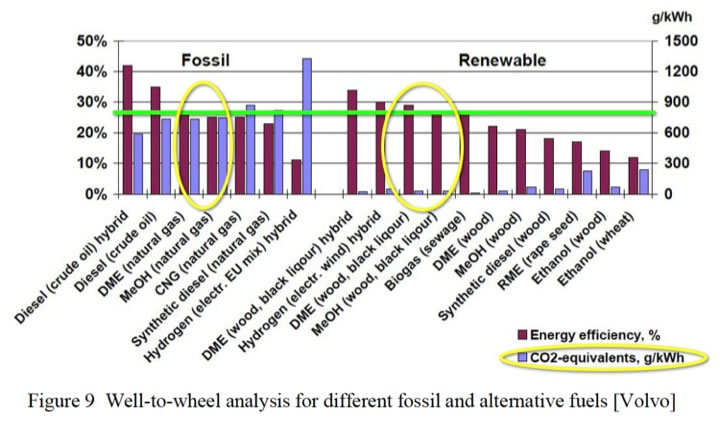
A quick look at the Wheel-to-Well CO2 emissions from various fuels will give us a picture of the wide range of possibilities in our transportation fueling picture. The California Air Resources Board (CARB) has complied this chart. It is the most complete info to date. As we develop the discussion below we will refer to this chart. Another way to look at GHG is to measure alternative fuels made from fossil feedstocks vs. alternative fuels made from renewable feedstocks. Note the much lower CO2 when methanol is made from renewables (yellow circles). This is a EU perspective.
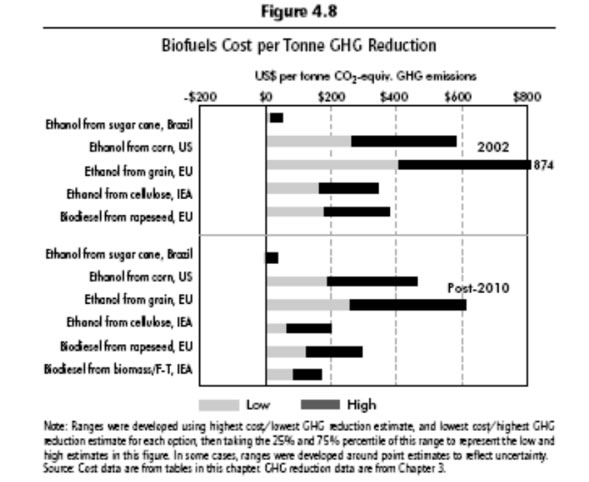
On a worldwide perspective, the chart below shows the cost of biofuels per ton of GHG reduction. Here it is clear that Brazilian sugarcane ethanol has a large advantage.
3. Electricity and Plug-In Hybrids
Plug-ins are Internal Combustion Engine Vehicles with electric assist. They are assisted by batteries for a limited number of miles, and then the batteries are recharged, both by the engine/braking system, and by plugging them into an outlet. They have a major advantage over pure electric vehicles because when the battery is discharged, they can run on their fuel. As we will propose, methanol/ethanol fueled Plug-ins, or even conventional hybrids, represent a very good compromise, that reduces fuel use and overall conserves energy, if the electricity used to charge them is not contributing more CO2.
4. Alcohol Fuels
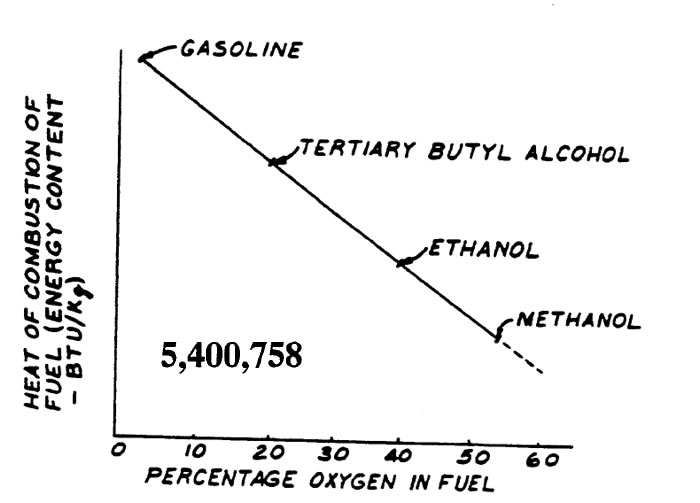
Alcohols burn differently than gasoline. The heat of combustion of 3 alcohols and gasoline is show below.
4.1. Ethanol Fuel
With the US ethanol fueling mandate of 36 billion gallons by 2022, making ethanol in every way possible will be necessary. The pro and cons of ethanol are discussed here. Although it is primarily made from corn in the US, we have now discovered a way of making it from natural gas. Most recent studies have shown a positive energy balance for ethanol made from corn to be between 23 and 40 percent.
4.2. Methanol Fuel
Methanol Fuel was used in California in State government tests for over 15 years (see California methanol fuel experience ). It is used in racing because engines can get a lot of power out of it. In the early 1990s through Ford's in-house research, and the demonstration fleets in the field, the technology for methanol-fueled vehicles was brought to a high level of reliability - high enough for Ford to make the decision to take this technology to production. It is a fuel that we can make from coal, natural gas, or any organic wastes we have. In fact, many people do not realize that methanol can be a biomass fuel also, probably without the need for a subsidy, as ethanol has had. Methanol is more easily produced from cellulosic or woody material than ethanol. In 1927, methanol was known as "wood alcohol" because it was produced via the destructive distillation of wood residues in the forests. It was cheaper to transport the methanol than it was to transport the wood.
It is seeing growing use in China, as we shall discuss later.
As we will point out later, it is also an excellent hydrogen carrier for the fuel cell cars of the future.
4.3. Methanol vs. Ethanol
According to Roberta Nichols, the Ford engineer that is credited with innovating the Flex Fuel vehicle; "These two alcohols actually complement each other. The performance and emissions of the two in an internal combustion engine are quite similar. Ethanol does not have a flame visibility issue like methanol (and methane and natural gas), because ethanol has two carbon molecules to form soot. This creates the yellow color in a bum. But ethanol also has a more difficult cold start because of the much lower vapor pressure (2.3 psi compared to 4.6 psi for methanol). Both alcohols have a single boiling point and a high latent heat of vaporization, which adds to the cold start problem, particularly at temperatures much below 8 °C (45 F). If the economics of the two alcohols were equivalent, ethanol would be the alcohol of choice for transportation use because the volumetric energy density is better, making on-board storage of the fuel less of an issue for the packaging engineer. But the economics of methanol are more favorable, making it the better choice for replacement of petroleum-based fuels in the transportation sector where the consumer is very much aware of the cost of fuel at the pump. The good news is, the FFV can use either methanol or ethanol and, in fact, some of the early experimental cars ran well on a combination of all three fuels (methanol, ethanol, and/or gasoline), which made them really flexible!"
4.4. Alcohol Blend Fuels - GEM Blends
The fact that both ethanol and methanol blend well with gasoline, enable a transition path of more and more alcohols to be added to our gasoline. The modern computers that determine engine running characteristics needed to utilize the blend in the best way are already in modern vehicles, and if necessary should be given a small change in the programs within them to accommodate the blends. We will discuss in some detail the use of blends in the coming Alcohol Blend Fuels section.
In our section on Lotus Testing of gasoline/ethanol/methanol fuel blends (called GEM blends, for example G28.8, E42.5 and M28.7 means 28.8% gasoline, etc.), we will see that there is an amazing flexibility available to us. For example the plot below shows the wide range of GEM Blends that will all perform exactly the same as E85, with the exception that the higher methanol blends output lower emissions.
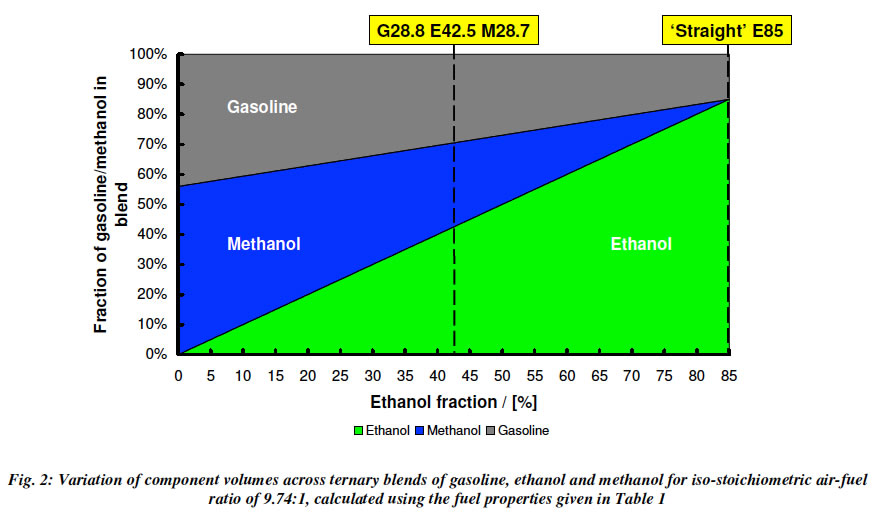
Turner et al. 2011 GEM Ternary Blends: Removing the Biomass Limit.SAE 2011-24-0113.
5. The Mobil Process for Making Gasoline from Methanol
The process first makes methanol and then dries it into Dimethyl Ether and then passes it over a catalysts ZSM-5 to wind up with some fraction of gasoline. The table below shows the outputs.
 Basically, the gasoline returned is .4366 x.85 = .3711. This means that the overall energy efficiency of a process that starts using natural gas to make methanol and then makes gasoline is ~ .70 x .3711 = .25 or 25%, because the efficiency of natural gas to methanol is ~ 70%. This is not the best use of raw materials, especially when methanol itself can run an internal combustion engine with higher efficiency and lower emissions.
Basically, the gasoline returned is .4366 x.85 = .3711. This means that the overall energy efficiency of a process that starts using natural gas to make methanol and then makes gasoline is ~ .70 x .3711 = .25 or 25%, because the efficiency of natural gas to methanol is ~ 70%. This is not the best use of raw materials, especially when methanol itself can run an internal combustion engine with higher efficiency and lower emissions.
more later
6. The Fischer-Tropsch Process for making Gasoline and Diesel fuels
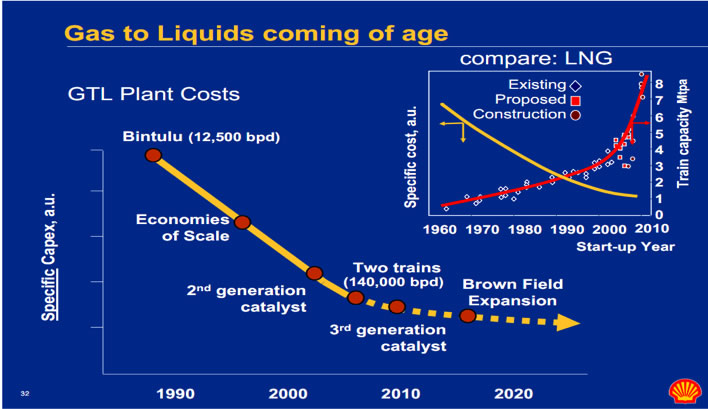
There are currently massive operations around the world that are producing diesel fuel and other hydrocarbons using the Fischer-Tropsch (F-T) process. Shell and Sasol are the leaders in this area. As the plot below shows the costs have undergone significant development, essentially over many decades, and cannot be expected to be reduced very much further. The costs of the resulting fuel is...
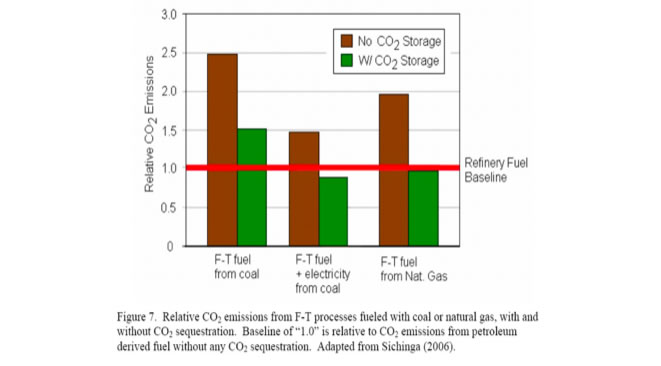
The other big problem with F-T is the CO2 emissions levels. As the graph below shows, looking at the brown bars which don't fantasize the possibility of CO2 storage and sequestration, we see that F-T from coal is 2.5 times as CO2 intensive as gasoline (the red line). F-T fuel from natural gas is almost 2 times as dirty as gasoline to make. The bar in the middle is a partial way to lower the CO2 emissions, by using the F-T process heat output to generate electricity. This concept, in general, is a good idea no matter what the process is. But even with this combined process F-T is generating 50% more CO2 than the production of gasoline does.
More later.
7. Natural Gas
Natural gas has its place in the scheme of transportation, but when the advances in alcohols is fully recognized, it may be phased out even in these applications. See our analysis of natural gas as a transportation fuel.
8. Hydrogen
On paper, hydrogen is a good fuel, which emits little pollution. In reality we will briefly summarize the reasons why it will not be practical for many decades to come. See our analysis of hydrogen as a transportation fuel.
This page introduces pages on Electricity (Plug-ins), Ethanol, Methanol, Natural Gas, Bio-diesel and Hydrogen
.